Recently, the Drug Administration of Vietnam under the Ministry of Health issued an official document on administrative sanctions against BB Vietnam Trading Service Company Limited, headquartered in Hanoi. The decision was signed by Deputy Director General of the Drug Administration of Vietnam Ta Manh Hung on April 21, after identifying serious violations in the company’s cosmetic business operations. The total fine was set at 75 million VND, accompanied by strict remedial measures to ensure the rule of law in pharmaceutical and cosmetic management.
Details of the violation and legal basis
The main violation of BB Vietnam Trading Service Company Limited was identified as trading cosmetics without having or failing to present the Product Information File (PIF) within the prescribed time limit upon request for inspection by competent authorities. This is a mandatory legal dossier for every cosmetic product circulating on the market, containing information on safety, efficacy, and quality standards.
The Drug Administration of Vietnam affirmed that this behavior violated the provisions of point b, clause 3, Article 71 of the Government’s Decree No. 117/2020/ND-CP dated September 28, 2020, on administrative sanctions in the health sector. At the same time, this violation was also cross-referenced with the amendments and supplements in point b, clause 21, Article 2 of Decree No. 124/2021/ND-CP dated December 28, 2021. The lack of a PIF not only hinders post-inspection work but also poses risks in controlling the origin and safety of products for consumers.
Aggravating circumstances and the list of recalled products
During the handling process, the Drug Administration of Vietnam applied aggravating circumstances to the enterprise. The reason is that at the same time of inspection, up to 37 cosmetic products declared by the company committed the same violation regarding legal dossiers. This is clearly specified in clause 5, Article 71 of Decree No. 117/2020/ND-CP and point c, clause 21, Article 2 of Decree No. 124/2021/ND-CP.
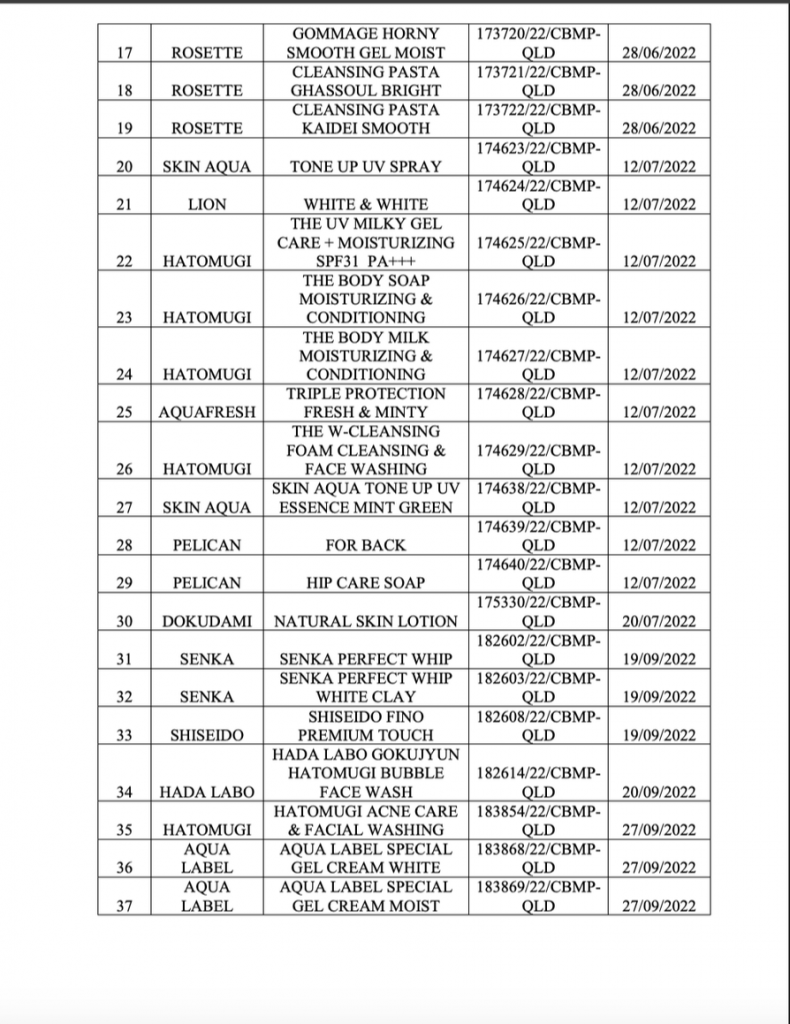
The list of 37 products forced to be recalled and destroyed includes many popular cosmetic brands with a large market share in Vietnam. Notably, this list includes names such as Hatomugi, Rosette, Skin Aqua, Senka, Shiseido, Hada Labo, and Aqua Label. In particular, the sunscreen product Anessa – which is widely trusted by consumers – is also subject to recall. However, it should be noted that this recall decision stems from a legal dossier violation (missing PIF), not from conclusions about poor quality or direct safety hazards for users at the time of inspection.
Remedial measures and the enterprise’s responsibility
Besides the monetary fine, the Drug Administration of Vietnam required BB Vietnam Trading Service Company Limited to take responsibility for coordinating with relevant agencies and units to recall and destroy the entire quantity of the 37 aforementioned products. The enterprise is obliged to report the implementation results to the Drug Administration of Vietnam within 30 days from the effective date of the decision.
The sanction decision takes effect immediately from the date of signing. The enterprise must pay the fine to the state budget within 10 days; if the deadline is exceeded, enforcement measures will be applied according to regulations. In addition, the law also reserves the right for the enterprise to file complaints or initiate administrative lawsuits if there are grounds to disagree with the management agency’s decision. This is a strong warning for cosmetic business units to fully comply with administrative procedures and store product information files in accordance with the regulations of the Ministry of Health.
Below is the list of recall product:
Center for Support & Continuing Education